Drug delivery systems are evolving rapidly to improve stability, safety, and therapeutic outcomes. Among these innovations, dry powder injectable formulations are gaining attention for their role in overcoming limitations of traditional liquid injectables. By using non-aqueous formulations, pharmaceutical science is redefining how injectable drugs are developed, stored, and administered.
This blog explores what dry powder injections are, why they matter, and how they are transforming modern drug delivery.
What Are Dry Powder Injections?
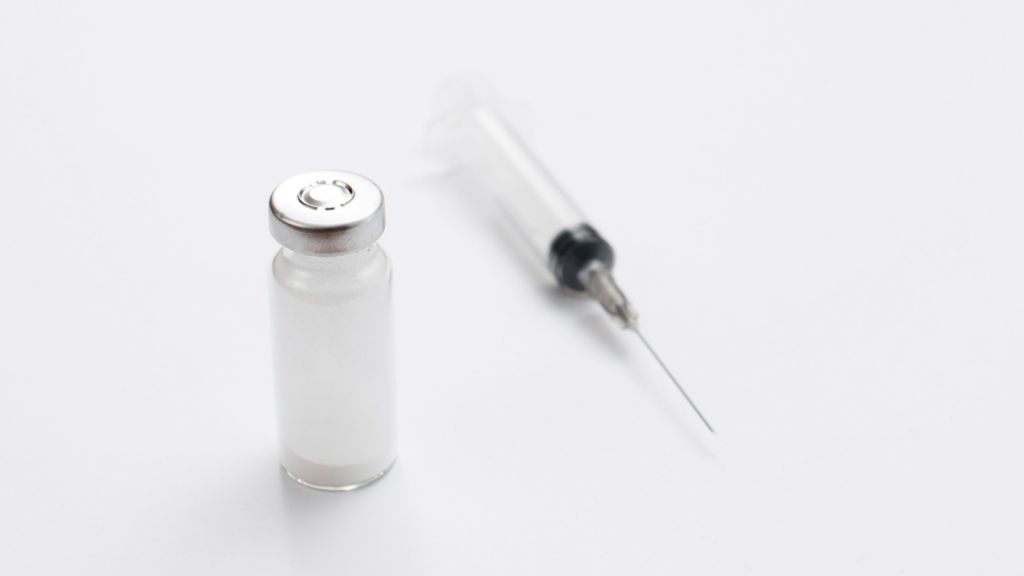
Dry powder injections are injectable medicines supplied in a solid, powder form rather than as a liquid solution. Before administration, the powder is reconstituted with a suitable sterile diluent to form an injectable solution or suspension. Unlike conventional liquid injectables, dry powder injections do not contain water during storage. This non-aqueous nature plays a key role in improving drug stability and shelf life.
The product range of dry powder injections at Kilitch Drugs India Ltd. includes:
- Ceftriaxone Powder for Injection (500mg/1g)
- Cefepime Powder for Injection (1g)
- Meropenem for Injection (500mg/1g)
- Caspofungin Acetate for Injection (50mg/70mg)
- Piperacillin + Tazobactam Injection (2.25gm/4.5gm)
These products are designed to ensure stability and reliability, delivering high-quality care in treatment.
Why Non-Aqueous Formulations Matter
Many active pharmaceutical ingredients are unstable in water. Exposure to moisture can lead to chemical degradation, reduced potency, or microbial growth. Non-aqueous formulations help overcome these challenges by eliminating water during storage.
By keeping the drug in a dry state, manufacturers can preserve its chemical integrity for longer periods. This is especially important for antibiotics, biologics, and other sensitive compounds.
Improved Stability and Longer Shelf Life
One of the biggest advantages of dry powder injections is enhanced stability. Drugs that degrade quickly in liquid form can remain stable for years when stored as a powder. This longer shelf life reduces wastage, improves supply chain efficiency, and ensures consistent drug quality. It is particularly beneficial in regions with limited cold-chain infrastructure.
Role of Lyophilisation in Dry Powder Injections
Most dry powder injections are produced using a process called lyophilisation, or freeze-drying. In this process, the drug solution is frozen and then dried under vacuum to remove water without damaging the active ingredient. Lyophilisation helps maintain the structure and activity of complex molecules, making it ideal for injectable drugs that are sensitive to heat or moisture.
The product range of lyophilized injections at Kilitch Drugs India Ltd. includes:
- Azithromycin Powder for Injection (500mg)
- Omeprazole for Injection (40mg)
- Pantoprazole for Injection (40mg)
These lyophilized products ensure stability and effectiveness, even for heat-sensitive medications.
Faster Reconstitution and Accurate Dosing
Modern dry powder injections are designed for quick and easy reconstitution. When mixed with the recommended diluent, the powder dissolves or disperses rapidly, forming a uniform injectable solution. This ensures accurate dosing and consistent therapeutic effects. Healthcare professionals can administer the medication efficiently without compromising safety or effectiveness.
Applications in Critical and Specialized Therapies
Dry powder injections are widely used in antibiotics, oncology drugs, hormones, and vaccines. Many of these drugs require high bioavailability injectables and precise dosing to achieve desired outcomes. In emergency and hospital settings, dry powder injections provide reliable treatment options where rapid action and drug integrity are essential.
Advantages Over Liquid Injectable Formulations
Compared to liquid injectables, dry powder injections offer several advantages:
- Reduced risk of degradation
- Lower need for preservatives
- Improved microbial safety
- Better storage flexibility
These benefits make non-aqueous injectable formulations an attractive choice for both manufacturers and healthcare providers.
Challenges and Considerations
Despite their advantages, dry powder injections also present challenges. They require careful handling during reconstitution and strict adherence to preparation guidelines. Manufacturing dry powder injectables is also complex and cost-intensive. However, ongoing technological advancements are helping streamline production and improve ease of use.
The Future of Non-Aqueous Injectable Drug Delivery
The future of injectable drug delivery is increasingly focused on stability, precision, and patient safety. Non-aqueous formulations and dry powder injections align well with these goals.
As research continues, newer formulations with faster reconstitution, improved solubility, and enhanced delivery systems are expected to emerge. These innovations will further expand the role of dry powder injections in modern medicine.
Conclusion
Dry powder injections represent a significant advancement in injectable drug delivery. By leveraging non-aqueous formulations, they offer improved stability, longer shelf life, and reliable therapeutic performance. As healthcare demands grow more complex, these formulations are becoming essential in delivering high-quality injectable medicines.
Kilitch Drugs (India) LTD continues to focus on developing advanced dry powder injection solutions, supporting safer, more effective, and future-ready drug delivery systems.